Nitrous Oxide Decomposition Mechanism
How the N2O greenhouse gas is decomposed.
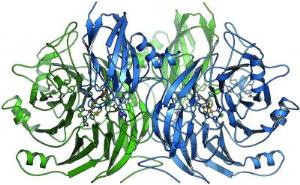
The N2O-reductase enzyme possesses four reactive centers for the decomposition of nitrous oxide into elemental nitrogen. [Source: BIOSS/ University of Freiburg]
Nitrous oxide (N2O) is a harmful climate gas. Its effect as a greenhouse gas is 300 times stronger than that of carbon dioxide.
Nitrous oxide destroys the ozone layer. In industrial agriculture, it is generated on excessively fertilized fields when microorganisms decompose nitrate fertilizers. Decomposition of nitrous oxide frequently is incomplete and strongly depends on environmental conditions.
Researchers from Freiburg, Constance, and KIT have now identified the structure of the enzyme that decomposes nitrous oxide and the decomposition mechanism.
Their results are published in the Nature journal [see below].
The study demonstrated that the N2O-reductase enzyme possesses active centers made up of four copper atoms and two sulfur atoms. "Surprisingly, we found that microbiologists all over the world have assumed an incorrect structure so far," explains Professor Oliver Einsle, group leader at the Institute of Organic Chemistry and Biochemistry of the University of Freiburg. Scientists have assumed a single sulfur atom only and have not been able to completely identify the nitrous oxide decomposition mechanism. Based on the new data, the reaction sequence of the enzyme can be modeled much better. Future investigations are to provide further details and help understand which influence environmental conditions have on the process.
"It was of decisive importance that all steps of our investigation were executed in the absence of air oxygen," emphasizes Walter G. Zumft, retired professor of Karlsruher Institute of Technology. In contact with oxygen, parts of the enzyme react and the enzyme changes its structure. Together with Dr. Anja Pomowski from the University of Freiburg, the bacteria were cultivated under an oxygen-free atmosphere, the enzymes were isolated on a large scale, crystallized, and the structure was analyzed using X-rays. The team of four authors was completed by Professor Peter Kroneck from the University of Constance.
"The current study provides interesting and complementary insight into the nitrogen cycle," says Dr. Ralf Kiese from the KIT Institute of Meteorology and Climate Research. Nitrous oxide and nitrogen production on fields, pastures, and in forests depends on a multitude of often opposing effects. Last year, a KIT study demonstrated that animal husbandry may lead to less nitrous oxide unter certain conditions (see below). Detailed knowledge of microbial processes and their dependence on environmental conditions might help to better model the nitrous oxide contribution to the climate. In the long term, it might even be feasible to use the knowledge in order to prevent nitrous oxide from being released into the atmosphere, for example, by additives in fertilizers that preserve the functioning of N2O-reductase or by optimized processes in sewage treatment plants.
Karlsruhe Institute of Technology: KIT is a public corporation according to the legislation of the state of Baden-Württemberg. It fulfills the mission of a university and the mission of a national research center of the Helmholtz Association. KIT focuses on a knowledge triangle that links the tasks of research, teaching, and innovation.
Further Information:
Article published: 23.08.2011
Anja Pomowski, Walter G. Zumft, Peter M. H. Kroneck, and Oliver Einsle:
N2O binding at a [4Cu:2S] copper-sulphur cluster in nitrous oxide reductase.
In: Nature; published online 14 August 2011, DOI 10.1038/nature10332
Benjamin Wolf, Xunhua Zheng, Nicolas Brüggemann, Weiwei Chen, Michael Dannenmann, Xingguo Han, Mark A. Sutton, Honghui Wu, Zhisheng Yao, Klaus Butterbach-Bahl:
Grazing-induced reduction of natural nitrous oxide release from continental steppe.
In: Nature; 464, 881-884, 8 April 2010, DOI 10.1038/nature08931
Quelle: Karlsruhe Institute of Technology, KIT, Germany
Citation: http://www.internetchemistry.com/news/2011/aug11/nitrous-oxide-decomposition.html
(C) 1996 - 2016 Internetchemistry







